December 1, 2001
Introduction of the new TaXi software system is proceeding remarkably smoothly. As expected, there were numerous minor problems but no major obstacles. We are happy to see that many centers have been able to install TaXi and are now in possession of a functional local transplant data base.
As stated from the outset, use of TaXi is not mandatory for CTS participants. However, even those who wish to continue reporting on paper (CTS forms or printouts) can utilize TaXi by receiving periodic data downloads from us by e-mail. The TaXi package, including your center's data, can be loaded onto a local PC making it possible to perform statistical analyses using standard software packages. If you would like to make use of this option, please do not hesitate to contact us.
We have received several useful suggestions for the improvement of TaXi. If you have ideas about how the program could be further improved, we would be grateful for your suggestions.
With the support of 29 laboratories and transplant centers, we were able to collect pretransplant sera from 4000 kidney graft recipients. You may recall the report on anti-Fab antibodies in a previous CTS Newsletter (3:1999). We are now pleased to report on an extensive analysis of IgG antibodies directed against HLA antigens in these sera, and the influence of these antibodies on subsequent kidney graft survival. The special feature of the study is the use of an advanced ELISA technique, which allows differentiation of anti-HLA class I from anti-HLA class II antibodies. Results obtained in smaller transplant series, using similar techniques, have been published in the literature and are in part conflicting.
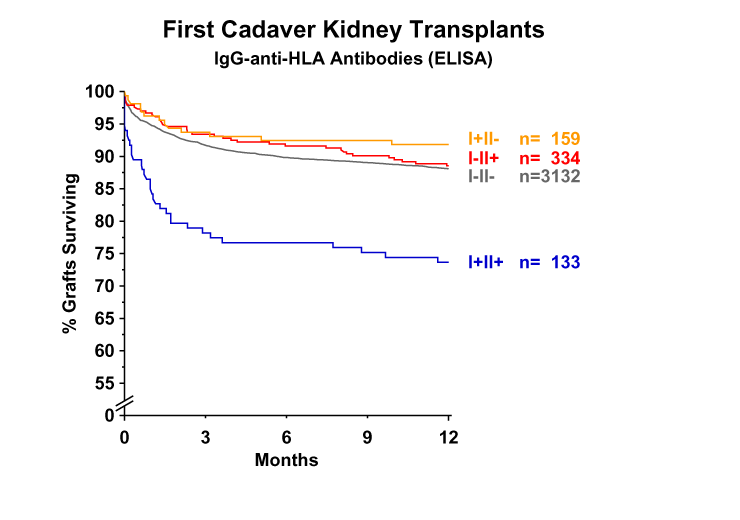
As shown in Figure 1, when first cadaver kidney transplants were analyzed, patients who had antibodies reactive against both HLA class I and HLA class II in their pretransplant serum (I+ II+) had a significantly lower graft survival rate than patients who possessed antibodies against only HLA class I (I+ II-) or only HLA class II (I- II+). The latter two groups were not statistically different from patients who showed no evidence of antibody reactivity at all (I- II-).

This surprising finding was shown not to be the result of better HLA matching for class I or class II antigens in the antibody I+ II- or I- II+ groups. In other words, even when HLA mismatches were present, sensitization against either HLA class I or HLA class II had no deleterious effect. In contrast, among the I+ II+ patients, those with a good HLA match (0 or 1 mismatch for HLA-A+B+DR) had an excellent graft outcome (87% at 1 year), whereas those with 4-6 mismatches had a 1-year graft survival rate of only 72%.
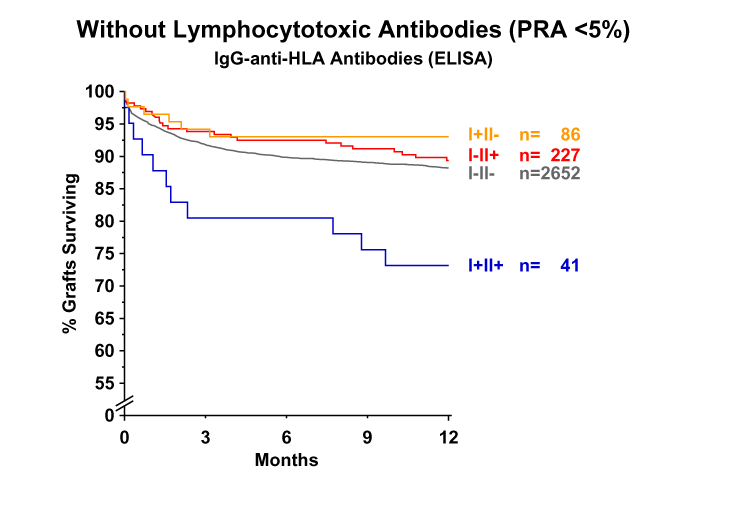
As shown in Figure 2, the analysis in patients without preformed lymphocytotoxic panel-reactive antibodies (PRA <5%) showed a result similar to the overall series. Moreover, in patients in whom preformed lymphocytoxic antibodies were found (PRA ?5%), the deleterious effect of presensitization was also limited to the I+ II+ group (Figure 3). It therefore appears that deleterious I+ II+ antibodies can be found in patients with no evidence of PRA in the conventional antibody screening assay, and that among patients who showed evidence of preformed antibodies in the PRA test, only those who possessed antibodies reactive against both HLA class I and II were adversely affected.
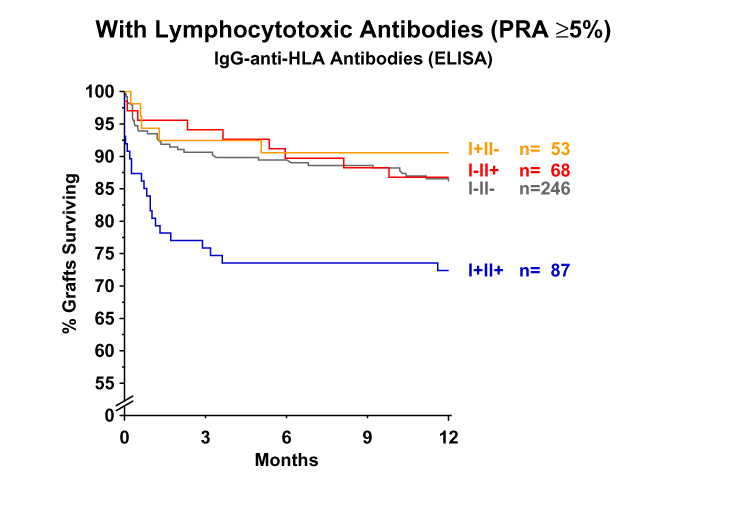
Another additional and very important finding was that, whereas some patients without PRA could be shown to possess ELISA reactive I+ II+ antibodies, there was another patient group of equal size which showed reactivity in the PRA test but was completely negative when tested by ELISA. Importantly, the antibodies detected solely in the PRA assay were clinically relevant ! The graft success rate in these patients was only 74% at 1 year.
Altogether, these results show that one cannot simply replace PRA testing with ELISA testing, or vice versa. Additional useful information can be derived from the use of both antibody assays. We are planning to continue this work with the goal of developing an informative but cost-effective strategy for antibody testing of patients prior to transplantation. Since the number of patients within the most informative groups is very small, one goal of our future work will be to expand the study to an even larger number of transplants. Consequently, we will be contacting CTS participating laboratories by separate mail in order to ask for their support.
Thanks are due to the laboratories and clinical transplant centers who contributed sera and data for the study:Barcelona, Spain (Dr. Martorell); Berlin, Germany (Dr. Schönemann); Budapest, Hungary (Dr. Padanyi); Cardiff, United Kingdom (Dr. Rees); Dallas, USA (Dr. Stastny); Freiburg, Germany (Dr. Lang); Geneva, Switzerland (Dr. Jeannet); Glasgow, United Kingdom (Dr. Farrell); Heidelberg, Germany (Dr. Wiesel); Helsinki, Finland (Dr. Koskimies); Kentucky, USA (Dr. Thompson); Lausanne, Switzerland (Dr. Aubert); Leicester, United Kingdom (Dr. Horsburgh); Liege, Belgium (Dr. Bouillenne); Ljubljana, Slowenia (Dr. Bohinjec); Mannheim, Germany (Dr. Schnülle); Marburg, Germany (Dr. Lange); Munich, Germany (Dr. Scholz); New York, USA (Dr. Fotino); Phoenix, USA (Dr. Vyvial); Prague, Czech Republic (Dr. Ivaskova); Quebec, Canada (Dr. Roy); Reims, France (Dr. Cohen); Rijeka, Croatia (Dr. Vujaklija-Stipanovic); Rio de Janeiro, Brazil (Dr. Goncalves De Freitas); Strasbourg, France (Dr. Tongio); Sydney, Australia (Dr. Doran); Zagreb, Croatia (Dr. Kastelan)
All graphs shown on the CTS website were updated at the end of November. Thank you for providing your clinical updates in time for the generation of these new statistics. Naturally, the center-specific analysis function remains active and is available to all CTS participants, regardless of whether they use TaXi or not.
One month ago you received the yearly request for confirmation of data on malignant tumors in your patients. Please assist us with the important task of generating accurate cancer statistics as a basis for attempts to reduce the future incidence of cancer in transplant recipients. In order to reduce the likelihood of underreporting, centers are included in the analysis of cancer data only if the questionnaire is returned. Therefore, please complete and return the blue "Cancer Confirmation Questionnaire" at your earliest convenience. A useful hint: routinely asking for the presence of malignancies when compiling information for your quarterly CTS updates will greatly simplify the yearly confirmation of cancer data.
Thank you for your continued support of the international transplant study
Sincerely yours,