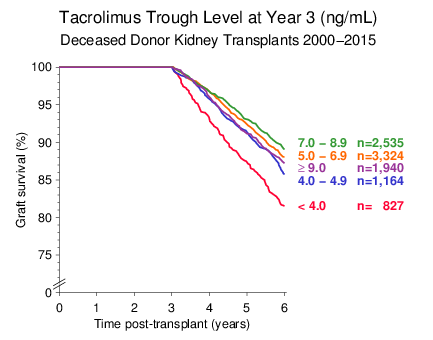
Figure 1
CTS Collaborative Transplant Study
Dear Colleague
We reported previously that a too low tacrolimus trough level <5 ng/mL at year 1 after kidney transplantation is associated with significantly impaired graft survival (CTS Newsletter 1:2014). In a recent AJT publication we analyzed the impact of tacrolimus trough levels and their fluctuations at post-transplant years 1, 2, and 3 (Süsal, C and Döhler, B. Late intra-patient tacrolimus trough level variability as a major problem in kidney transplantation: A Collaborative Transplant Study Report. Am J Transplant, 2019 [Early View] doi:10.1111/ajt.15346).
In patients with a functioning graft more than 3 years, the critical threshold for the tacrolimus trough level at year 3 was 4 ng/mL, slightly lower than the critical 5 ng/mL level at year 1. This effect is illustrated in Figure 1 for deceased donor kidney transplantations performed during 2000 2015.

Figure 1
Besides a too low tacrolimus trough level, intra-patient variability (IPV) of trough levels, especially during the first year after transplantation, was claimed to be a useful predictor of outcome. In the AJT paper we analyzed the impact of IPV in later phases of transplantation. We first defined IPV as the variation coefficient which is the ratio between standard deviation and arithmetic mean of trough levels measured at years 1, 2, and 3 post-transplant. An IPV of ≥30% was present in almost one-third of patients and was associated with a strongly impaired outcome (Figure 4 of AJT paper).
A simple method for IPV calculation is building a ratio between two trough levels. By merely considering the trough levels at years 1 and 2 and dividing the larger by the smaller value, IPV ratios could be calculated that were strongly associated with subsequent graft survival up to year 6 (Figure 2). In the multivariable analysis, the risk of graft loss was 29% higher in patients with an IPV of 1.51.9 and 82% higher in patients with an IPV of ≥2.0.
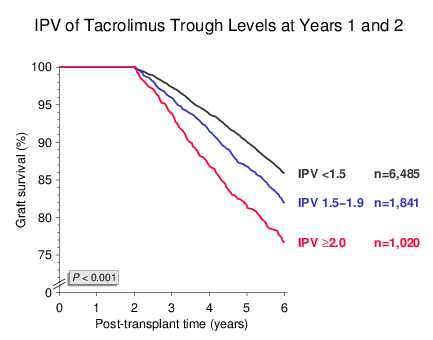
Figure 2
Similar results were obtained when IPV was calculated as the quotient of trough levels obtained at years 2 and 3 post-transplant (Figure 3). Again, almost one-third of patients with a functioning graft at year 3 had an IPV of ≥1.5 and showed impaired outcome in subsequent years. IPV had a strong impact on death-censored graft survival whereas its influence on patient survival was less pronounced (not shown).
As illustrated in Figure 4, it made no difference whether the reason for IPV was a decrease or increase; both a ≥1.5-fold higher decrease as well as increase of the trough level from year 2 to 3 was associated with a significant deterioration of graft survival beyond year 3 (as compared to IPV <1.5, P<0.001 for both).
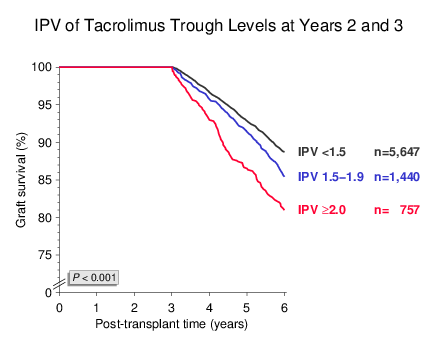
Figure 3
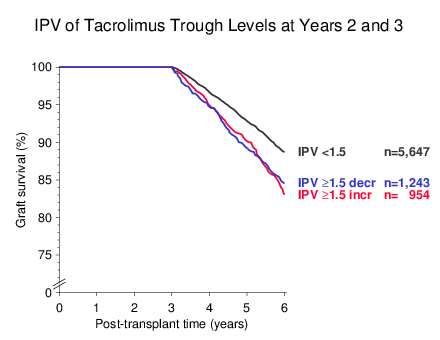
Figure 4
Another interesting aspect was the strong interaction of recipient age with the IPV effect. When IPV was calculated as the quotient of trough levels at years 1 and 2, a remarkably strong influence of IPV ≥1.5 was observed in the pediatric population of <18-year-old recipients. In contrast, the IPV effect was less pronounced in ≥60-year-old recipients than in the other age groups (Figure 5). The multivariable Cox regression analysis confirmed these results with accordingly increased hazard ratios for graft loss of 2.13 in <18-year-old, 1.50 in 1859-year old and 1.28 in ≥60-year-old recipients (P=0.002, P<0.001 and P=0.036, respectively).
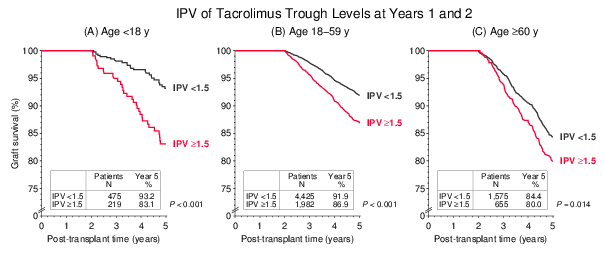
Figure 5
Our results altogether indicate that a high IPV of tacrolimus trough levels, also in the later post-transplant phase, is a major problem in kidney transplantation. Finding out the reason for fluctuation and eliminating it where possible is expected to result in further improvement of long-term outcomes in kidney transplantation.
The results presented in this Newsletter are another example of the excellent collaboration within the Collaborative Transplant Study. We are indebted to those of you who supplied us with this important information that is requested on our Immunosuppressive Follow-Up questionnaires. Please continue delivering this information using the recently updated CTS forms.
The next shipping date of Serum and DNA is
June 17/18, 2019.
Thank you very much for your continued support of the CTS.
Sincerely yours,

Caner Süsal